
Help Aisha Beat Brain Cancer
Donation protected
In early September 2022, just shy of her 30th birthday, our young and vibrant Aisha was diagnosed with one of the most aggressive and debilitating forms of brain cancer: diffuse midline glioma (DMG). She was given about 12 months to live. The diagnosis is typically considered to be a death sentence due to extremely aggressive growth of the tumour coupled with its dangerous location – the midbrain (it is essentially a glioblastoma, but specially categorized because of this). The midbrain is where functions such as breathing, heart rate, swallowing, movement, etc. are housed. It is also exceedingly rare, with an incidence rate of less than one in a million people, which in part limits the resources committed to finding a cure. And unfortunately, here in Canada, the medical system is limited in what it can offer, only offering a palliative approach.
However, cutting-edge scientific advances outside of Canada are very recently providing promising new treatments, and we are determined to leave no stone unturned to fight this cancer and give Aisha the best possible chance to live.
We have performed extensive research and consulted with international oncologists and researchers to construct a cocktail-type combination treatment plan over the next five years. In a number of instances, components of this approach have yielded complete remissions and 5+ year survivors (some of these treatments are so novel that we do not have endpoint data yet). In a situation like Aisha’s, when we are essentially told that survival is an impossibility, the reality that there are survivors makes our quest well worthwhile. Our objective is to do everything we can, as safely as possible, to give Aisha the best chance of being an outlier survivor. More specifically in instances of DMG, with only about 7,000 new cases per year, globally, every outlier survivor is meaningful (i.e. survival isn’t a one-in-a-million type of thing). There are lessons and methods to be learned and passed on. We are determined to make Aisha one of the survivors from whom others can leverage a cure for themselves or their loved ones.
Cancer is expensive to treat, and DMG is no exception, with the estimated minimum cost of drugs and therapies alone exceeding 1.1 million CAD. Our family will pool our resources to meet as much of the financial burden as we can; however, Aisha will need financial assistance beyond what we can put together in order to see the treatment through.
Those who know Aisha know of her vivacious spirit, infectious humour, and tenacity. Aisha has always followed her own path and passions in life.
She is a scientist, devoting her academic research to addressing global human-wildlife conflicts. Most recently, her work as a conservation biologist has been focused on species at risk in the Pacific Northwest (from the great orcas to the smaller sea snails). Aisha’s passion for animals is apparent through her beautiful photography and artwork of the wildlife she has devoted her career to protecting. She also rescued a stray and pregnant dog (Kaaya) from a rural dump site (who would otherwise have been euthanized) to give her a second shot at life, and in the process enriched five other families' lives with Kaaya's pups, with a reunion organized each year.

It is our earnest hope that this fundraiser will supplement our finances for the first two years of Aisha’s treatment. We will be constantly reassessing the situation as new treatments arise in the future. Due to the nature of the disease, with the endpoint occurring when the tumour grows such that it compresses the brain and compromises function, decisions have to be made and funded quickly.
If any funds are unutilized for this purpose, they will be donated towards research in the field to aid in the quest to find a cure for this tragic disease.
Aisha is being immensely brave facing this monumental and heartbreaking crisis. From being in a near-vegetative state for nearly two weeks, she bounced back to the amazement of her surgeon and oncologists. She has been an outlier ever since. The speed at which she regained her function, and the persistence of improvement through today continues to surprise all who are involved in her care. Her courage and resilience gives all of us the strength to travel along this journey with her. We firmly believe that she will continue to be a positive outlier, and we want to give her every opportunity to prove it.
We are asking for your kindness, generosity, and compassion in helping us if you have the means to do so, through donation and/or circulation of this initiative to friends and colleagues. We realize that this is a significant undertaking, and every gesture of assistance is invaluable to us, no matter how small it may seem to you.
We wouldn’t be asking for assistance if we did not believe we have a reasonable chance to save Aisha’s life through this effort. There is evidentiary backing to our belief, and scientific rationale for the mechanisms. We will detail this below, after the timeline, for the dual purposes of making it clear that we have a well-thought plan (rather than a “hope and pray” approach), and providing an educational framework for anybody who may be similarly affected and is interested in where Aisha’s journey has taken her.
We thank you from the bottom of our hearts for your generosity and support.
----------------------------------------
TIMELINE OF EVENTS
July 2022: Aisha noticed she was having short-term memory issues. This was casually attributed to work-related stress, compounded by her team shrinking significantly.
Late August 2022: While her memory continued to worsen, she started having diplopia (double-vision), followed a few days later by headaches, nausea, and an intense feeling of pressure in her head.
September 2, 2022: She went to the ER, where a CT scan showed a large mass in her head. She was immediately admitted and transferred to Vancouver General Hospital.

September 5, 2022: A subsequent MRI at VGH identified that the mass was 4.8 x 4.1 x 6.3 cm, originating from the thalamus, but with spread into the brainstem and basal ganglia. The tumour had grown to obstruct drainage of cerebrospinal fluid (CSF), which was creating the feeling of pressure and exacerbating the symptoms that drove her to the ER.
September 8, 2022: A ventriculostomy was performed to address the CSF drainage issue, and a biopsy was taken. Aisha started to feel better nearly immediately after the procedure.

September 19, 2022: We learned that the pathology report from the biopsy sample confirmed that Aisha had a Grade IV diffuse midline glioma. We were told that the only chance Aisha would have would be to have immediate debulking surgery (craniotomy) on September 22, to remove as much of the tumour as safely possible (the goal was 50-60%).
Due to the location of the tumour, this is a risky surgery, one that can never result in a complete resection (removal) due to the high probability of total debilitation caused by the removal of healthy brain tissue from these critical parts of the brain. However, surgery was a prerequisite for further treatment, as due to Aisha’s age and the tumour’s size, there was very little space available in her skull to support the expected inflammation that would be caused by radiation and/or chemotherapy.
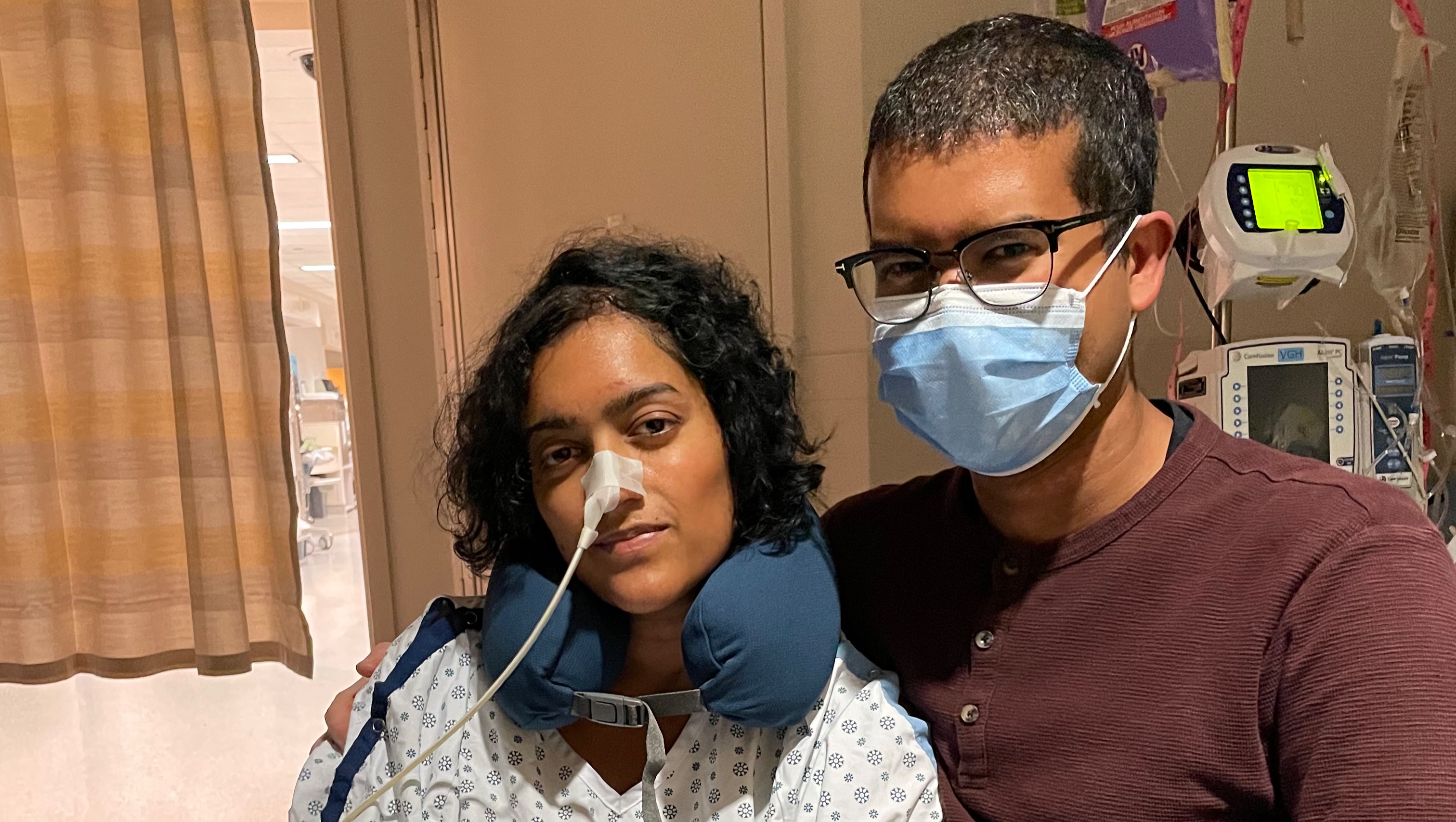
Aisha’s recovery from this 7-hour surgery was slow, and we were told that only “a small fraction” of the tumour had been removed. Aisha had lost her ability to speak, movement was limited to squeezing hands and wiggling toes, she was fed through a tube, catheterized, and in and out of consciousness.
September 28, 2022: We were told that if Aisha did not dramatically improve within 1-2 weeks, it would mean that the tumour’s growth was responsible for the entirety of her rapid debilitation, no physician would provide treatment to her, and no clinical trial would accept her due to her functionally disabled state (KPS of 30). At this point, she would only have weeks to live. We had just about lost all hope.

Remarkably, less than a week later, while still nonverbal and unable to stand without assistance, she had improved significantly enough that she was greenlit for radiation therapy.
Aisha continued to improve, regaining her speech, movement, memory, and cheerful demeanor, when radiation therapy commenced on October 17th. Chemotherapy was not administered concurrently due to concerns about her ability to handle inflammation due to the tumour’s increased size.
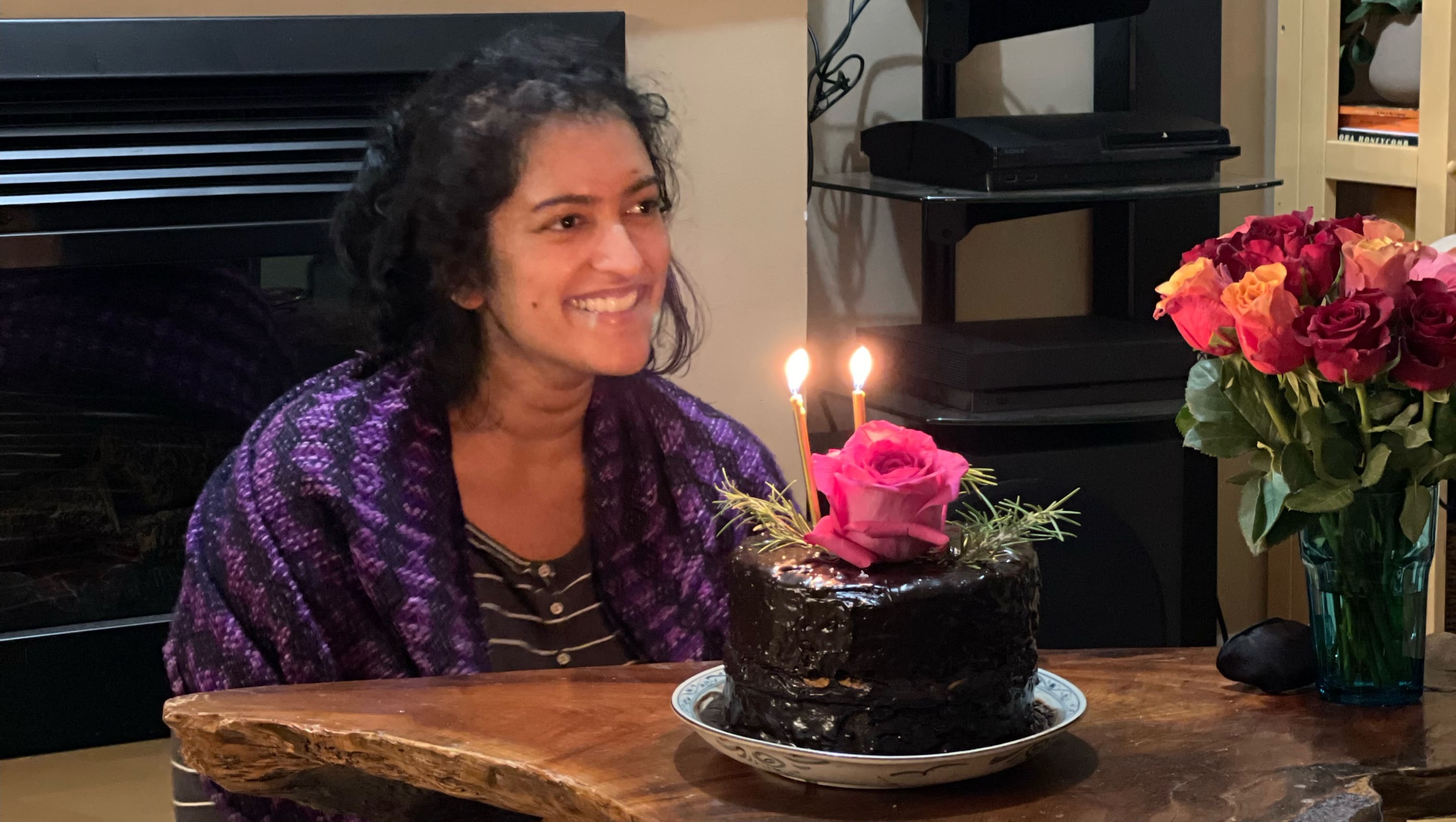
October 31, 2022: Aisha celebrated her 30th birthday at home, with friends and family - something that felt impossible just a month prior.
Throughout radiation, Aisha still continued to improve, defying the odds. She regained her fine motor skills and cognition to the point of being able to play piano and produce restaurant-calibre meals without assistance. Her memory also remarkably improved despite being exposed to 90% of the maximum radiation dose for the brain (54 Gy). Even more remarkably, Aisha’s diplopia completely resolved, with the only plausible explanation being that the brainstem portion of her tumour (at a minimum) had shrunk!
November 29, 2022: Radiation completed with no functional side effects, and only some hair loss to show for it. We could not believe how well Aisha was able to handle the treatment, not even having nausea (which we were told would be all but guaranteed). Her treating physician determined that she did not even need the powerful steroid (dexamethasone) nearly everybody in her position requires to combat the toll radiation takes on brain tissue.

Today, about four weeks post-radiation, we are continuing to give Aisha’s brain time to heal. While Aisha’s short-term memory is still deficient, it is slowly improving, and she is otherwise fully functional. It gives us and her physicians increased hope that she will be one of the outlier survivors we are striving for. Aisha has adopted dietary changes and is taking anti-inflammatory supplements, and will soon commence with experimental treatment in our quest to eradicate this tumour.
----------------------------------------
TREATMENT PHILOSOPHY
Cancer is an extremely complicated disease, compounded by countless variations of genetic mutations and other conditions that have material impact alone and/or in combination. While there can be many people diagnosed with a particular type of cancer, it is not unreasonable to suggest that within a cancer subtype, no tumour is identical.
All our research pointed to the fact that the most effective course of action to treat or cure Aisha’s cancer is a multi-modal one: combination therapy.
As such, we believe that the best method of attack is via multiple pathways: do as much as possible to insult the tumour from as many angles as possible, particularly as soon as it is safe to do so post-radiation, when the tumour is in its weakest state.
Unfortunately for Aisha, in Canada, treatment for DMG is only palliative in nature, focussed on a quality-of-life approach rather than a curative one. With this approach, even if the patient’s desire is to seek a cure rather than accept a 12-month prognosis, combining medication without evidenced safety through clinical trials is discouraged. Dr. Henry Friedman, a neuro-oncologist at Duke who assessed Aisha’s case, outwardly believes that “you’re curable until proved otherwise”; this is the approach that we are aligning with.
The problem in “playing it safe” with a single treatment is that this single treatment only attacks the tumour via one pathway. The tumour will inevitably mutate and develop resistance towards this drug. The tumour then starts to grow again (“recurrence”) along a different pathway, and this re-starts the countdown on the affected DMG patient’s life.
It is much more difficult to attack a growing tumour; as such, our objective is to assault the tumour from all possible fronts as soon as possible. Doing so while the tumour’s growth is arrested, and while the tumour has likely shrunk from radiation, increases the chances that we can induce further shrinkage, thus reducing the tumour cell count. This will accordingly reduce the number of cells that could conceivably mutate, and via this multi-pronged approach, mitigate an impactful mutation on any one pathway through a sustained attack on other pathways.
Despite the dismal prognosis of DMG, which is very similar to glioblastoma, there are long-term survivors. What we are finding is that one commonality they seem to share is that they took their treatment approach into their own hands, and aggressively pursued combination therapy.
A key advocate for this is Ben Williams, who has now survived for 28 years after taking ownership of his treatment: https://virtualtrials.org/surviveben.cfm
This approach has been documented in Surviving Terminal Cancer: https://www.youtube.com/watch?v=pJzpFrOCuMk
In consultation with Dr. Raymond Chang, a US-based oncologist featured in Surviving Terminal Cancer, we have put together a treatment plan for Aisha.
Some low-cost approaches Aisha has already adopted include ketogenic diet, intermittent fasting, off-label medication (Metformin, Celebrex, Montelukast), and an assortment of supplements, with special focus on anti-inflammatories.
However, the treatments with the most objective promise will incur a significant financial burden, beyond our limited resources. And so, Aisha and her family are making this appeal to you, and we would be eternally grateful to you for any assistance you could provide for her future treatment.
TREATMENT #1: ONC201
What is it, and why is Aisha a good candidate for it?
ONC201 is a new drug, currently in development, with astounding results for DMG. It is the only known compound with encouraging activity on tumours with the H3K27M mutation, which is the landmark DMG mutation. Interestingly, while the mutation is found in nearly all pediatric cases, it is only found in about 15% of adult cases. In this context, Aisha is extremely fortunate to have this mutation.
ONC201 has also shown to be more effective in adults than children.
Most compellingly, ONC201 has had dramatic success in thalamic DMGs, such as Aisha’s (https://academic.oup.com/neuro-oncology/article/22/Supplement_3/iii347/6018816):
- For non-recurrent patients, with median follow-up of 21.9 months (8.6–26.6) from diagnosis, median PFS or OS have not been reached. This surpasses historical OS of 13.5 months.
- This means that in nearly double typical median overall survival time, median recurrence hadn’t even been reached.
There are instances of patients surviving for 4+ years when treated with ONC201 as a monotherapy (with endpoint data not having been reached yet due to the novelty of the drug and associated trials).
ONC201 is presently in Phase II US clinical trials, with at least one Phase III trial greenlit for 2023. While there is scope for Aisha to be admitted into a US clinical trial, not only would there be risk that she would be assigned into a placebo control group, but also, she would be very limited in pursuing other treatments. As such, while the financial savings would be significant, we have a strong belief that even if we had certainty she would get ONC201 as a monotherapy, this would not give Aisha the best chance of sustained success. ONC201 is not available in Canada; however, it is available in Germany, where the identical compound has been manufactured and is available on a compassionate use basis.
Dr. Jürgen Arnhold, the prescribing oncologist in Germany, has about 400 H3K27M-mutant patients under his supervision, which represents more than the annual new cases in the US and France combined. He sees tumour shrinkage in about 33% of his patients, including a handful of complete remissions, with the best results in adults.
What is the cost (assuming stable exchange rates)?
ONC201 costs 265 Euros per capsule, with dosage of four capsules per week, potentially indefinitely depending on results. As such, we have to plan for this to be a recurring annual cost of 55,000 Euros, or about 80,000 CAD.
TREATMENT #2: ONC206
What is it, and why is Aisha a good candidate for it?
ONC206 is similar to ONC201, but stronger and with a mode of attack on a different pathway. This drug is newer, and less tested, but will be added to Aisha’s regimen in the event of either progression or shrinkage of her tumour. This will likely be determined after the results of her second post-radiation MRI, around June 2023.
What is the cost (assuming stable exchange rates)?
ONC206 costs 332.50 Euros per capsule, with dosage of two capsules per week, potentially indefinitely depending on results. As such, we have to plan for this to be a recurring annual cost of 35,000 Euros, or about 50,000 CAD.
TREATMENT #3: DENDRITIC CELL VACCINE
What is it, and why is Aisha a good candidate for it?
A dendritic cell vaccine is custom-made, created by harvesting the patient’s own dendritic cells and “priming” them to target tumour tissue for attack by T-cells.
A recently-reported Phase III trial for glioblastoma patients achieved positive outlier results (https://jamanetwork.com/journals/jamaoncology/fullarticle/2798847):
In pre-recurrent patients such as Aisha, 5-year survival was 13.0% with the vaccine vs. 5.7% without the vaccine.
While this vaccine typically requires a minimum 80% tumour resection and live tissue culture, neither of which Aisha has, she does have the H3K27M mutation, towards which a dendritic cell vaccine can be programmed to target. We intend to pursue this treatment via an institution in Germany: https://www.immune-therapy.net/en/spektrum/dc-therapie.php
The safety profile of this vaccine is exceedingly good: in nearly 2,200 administrations of the vaccine, only 5 serious adverse effects were noted to be possibly related (edema, nausea, lymph node infection).

What is the protocol for the vaccine?
Initially, Aisha will have to visit the institution in Germany, and undergo leukapheresis to harvest her dendritic cells. The vaccine will be developed in about a week, and the first dose will likely be administered there.
For the following three months, monthly doses will be administered in New York. For the following eight months, the vaccine will be administered every other month in New York.
In the second year, four doses will be administered in New York.
In the third year (and possibly fourth and/or fifth, depending on tumour state), three doses per year will be administered in New York.
What is the cost (assuming stable exchange rates)?
Exclusive of travel and accommodation, the vaccine cost for the first year will be about 75,000 Euros, or 110,000 CAD.
Second year cost would be about 55,000 CAD, and about 40,000 CAD thereafter.
TREATMENT #4: GAMMA-DELTA (γδ) T-CELL THERAPY
What is it, and why is Aisha a good candidate for it?
γδ T-cells are a first-line immune system response, seeking and destroying damaged and diseased cells identified with the presence of antigens not found in healthy human tissue.
γδ T-cells however only comprise about 0.5-5.0% of all T-cells; the aim of this therapy is to increase Aisha’s γδ T-cell count in concert with the administration of the vaccine, in order to maximize the attack on the tumour. This will be done by introducing pharmaceutical agents that dramatically multiply γδ T-cell count, using techniques such as this: https://journals.aai.org/jimmunol/article/175/3/1593/73049/Drug-Induced-Expansion-and-Differentiation-of-V-9V#R19.
This technique is in the very early stages of implementation and there is little in the way of published results. However, Dr. Raymond Chang of New York has been administering this therapy with success in his patients with similar profiles to Aisha.
The safety profile of this therapy is also very strong.

What is the protocol for this therapy?
It will be administered intravenously in New York, initially independently of the vaccine to rule out any non-combinatory adverse effects, and then in combination with the vaccine at each administration.
What is the cost (in Canadian Dollars, at current exchange rates)?
Each administration will cost about 7,000 USD, or about 10,000 CAD.
As such, the first-year cost would be about 80,000 CAD, dropping to 40,000 CAD in the subsequent year, and 30,000 thereafter.
FINANCIAL SUMMARY OF TREATMENT (CANADIAN DOLLARS AT CURRENT FX RATES)

It is our earnest hope that this GFM initiative will supplement our finances for the first two years. We will reassess the situation after that, as new, more effective, and more affordable treatments may become available in the future.
Thank you again; we are so grateful for your support.
----------------------------------------






Co-organizers (3)
Dmitry Uduman
Organizer
Calgary, AB
Elijah Willie
Co-organizer
Aisha Uduman
Co-organizer