- K
- D
- L
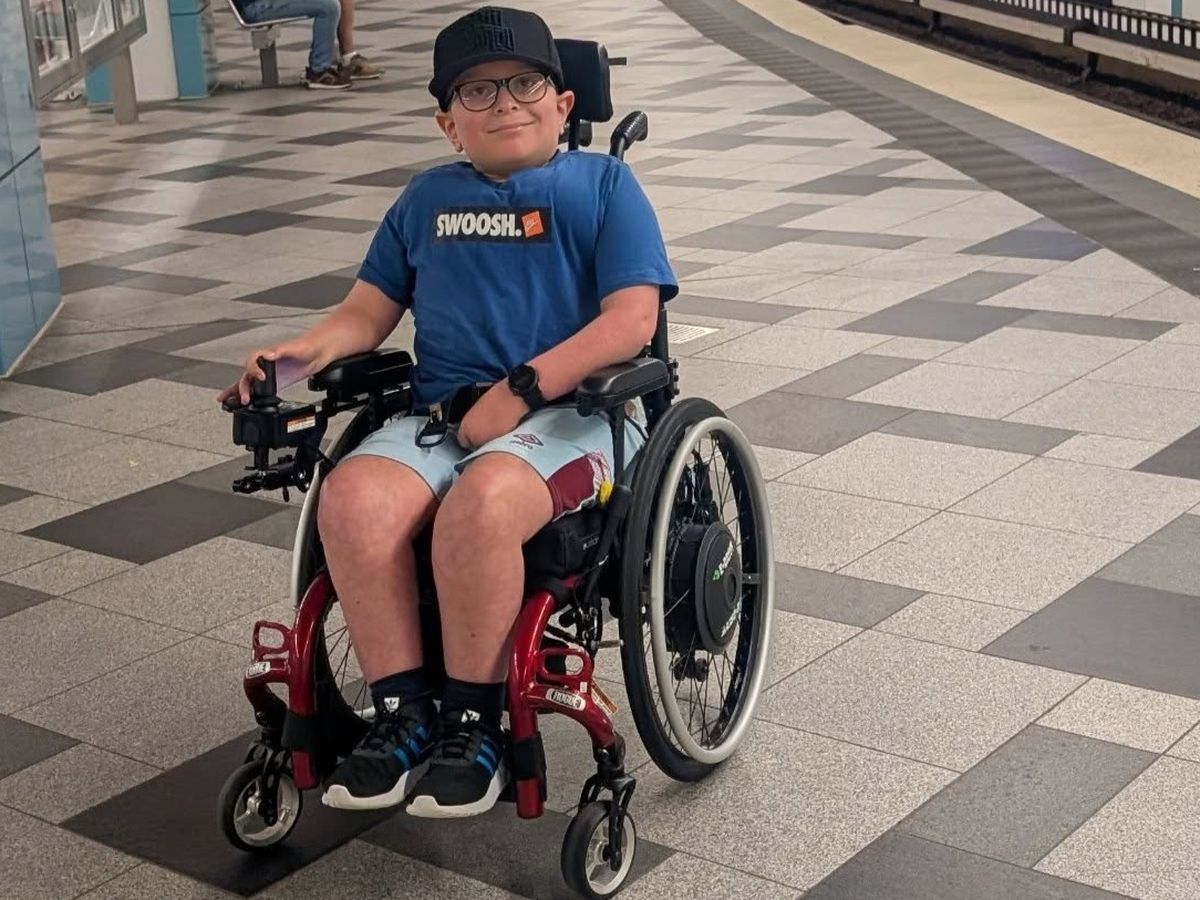
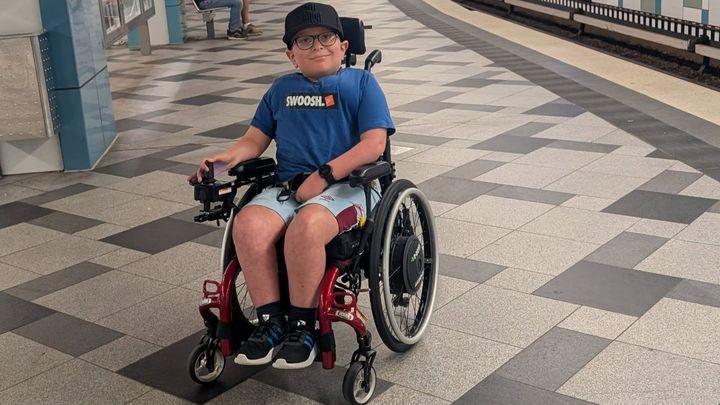
Meet Avery, our youngest son, a bright, kind, fun-loving 9-year-old boy, with a positive attitude and a big smile! In November 2017 at 18 months old, Avery was diagnosed with the rare genetic condition Spinal Muscular Atrophy (SMA) type 2. SMA is a rare, inherited neuromuscular condition. It causes progressive muscle weakness and muscle wasting.
Avery has never crawled, stood or walked and simple tasks like taking a pen lid off or getting dressed in the mornings are impossible for him to do alone. He requires extra support during the day, and his lack of strength leaves him unable to reposition himself at night, meaning his (and our) sleep is severely disrupted. It’s not uncommon to be called 5 or 6 times a night to reposition and get him comfortable.
As a consequence of the SMA, Avery also suffers from scoliosis as the muscles in his back are unable to support his spine. For this he wears a Spinal Jacket day to keep him upright and protects his internal organs from being compressed, which isn't the most comfortable, but unbelievably, he never moans.
When Avery was first diagnosed, there was no available treatment in the UK so we fought hard to get Avery onto a drug trial in Italy. He has been taking this drug now for almost 7 years and it's helped massively in terms of maintaining Avery's strength. We are even seeing small signs of strengthening which is amazing, but it is in no way a cure.
Life is very different for us as a family now, when we leave the house, we have to consider his needs first, will there be steps? Are toilets accessible? What equipment will we need? Will Avery's chair fit in? Is there a dropped kerb? Will Avery be included at parties or will he have to watch from the side?
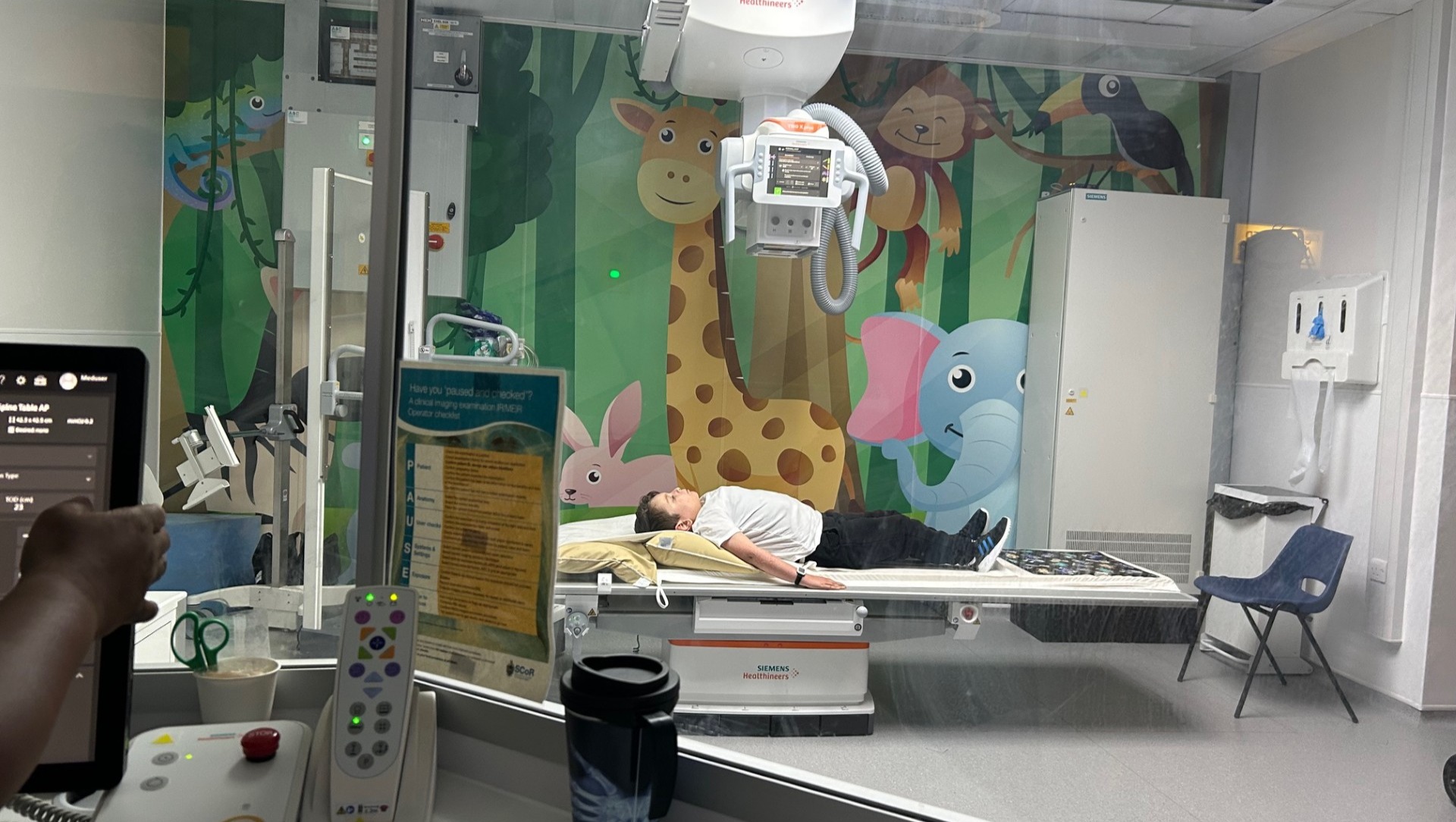
Inevitably Avery and his older brother Louis do miss out on a lot of the normal activities that boys do, for Avery even play dates at his friend's houses are rarely possible. Avery also has to attend numerous appointments at the John Radcliffe Children's Hospital, as well as weekly hydrotherapy and physio sessions.
But despite all this, he just brings sunshine into everyone's lives with his infectious smile, wicked sense of humour and never give up attitude! He has a huge talent and love of playing powerchair football and is the youngest competing in the national league, like most little boys, he dreams of playing for England one day!
Life for Avery is made easier with the right specialist equipment e.g. all-terrain wheelchair, hoist, non-slip mats and support such as physio and hydrotherapy - but unfortunately, these all come at a significant cost. We have had and continue to have amazing support from friends, family and our local community in helping raise funds for his treatment and equipment. Incredibly generous, kind people have organised a wide variety of events including horse shows, coffee mornings, dog shows, football matches, comedy nights, runs, walks and parachute jumps - all for our little boy, which has been just incredible!
This page has been set up to support their efforts. If you can help, we are truly grateful, your donation will help Avery have the best possible quality of life, to have fun and continue working towards an independent future.
Huge thanks,
Love,
Lisa & Alex x
Please note:
All funds raised are paid into an independently managed fund. Monies will only be drawn from this fund upon receipt of an invoice or signed purchase order that is directly related to equipment or treatment for Avery.














Organizer and beneficiary
A very special journey
Organizer
A Very Special Journey
Beneficiary